The Advantages of Differential pH Electrodes
Introduction
pH measurements are important in a variety of applications for understanding what is present in water, along with what reactions may occur in the solution. One example of this is in wastewater, where industrial processes and municipal systems must control pH within a specific range before releasing water back into the environment. Another use may be to monitor when a reaction occurs during a lab experiment.
These—and many, many other processes—use electrochemical pH electrodes that react to different pH levels in a predictable way (Nernstian), sending the electrical signal to a system for translation into something readable to humans (pH or mV). The most common type of pH electrode is a combination electrode, which uses two electrodes joined together to measure pH fairly accurately. They work well for most applications, but another type of electrode performs better in many circumstances: The differential electrode. The rest of this article covers the design differences between a standard combination electrode and a differential electrode, along with what performance benefits they provide.
Table of Contents
- Design Differences Between Standard and Differential Electrodes
- Differential Electrode Advantages
- Conclusion
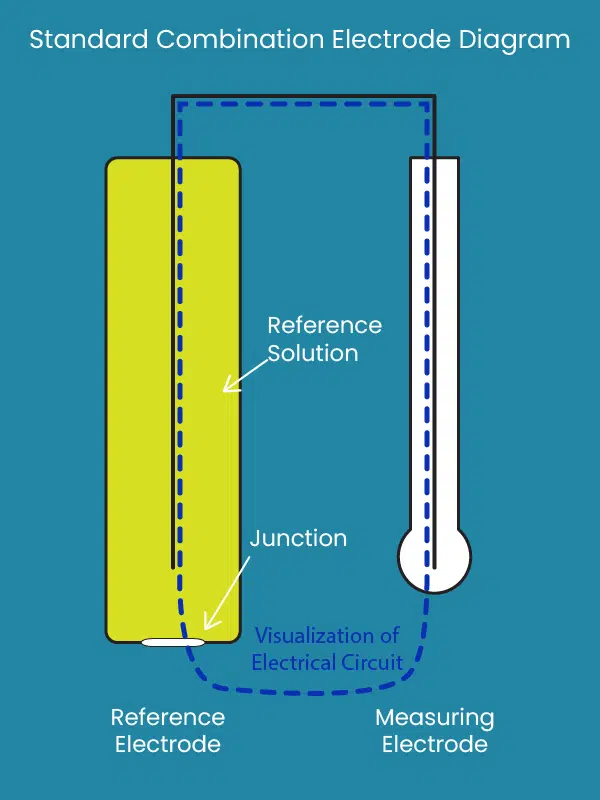
Standard Combination Electrode Design
To explain how a differential electrode provides better performance, we first must understand the differences between it and the standard electrode.
As mentioned in the introduction, most pH electrodes are combination electrodes. They mash together two electrodes into one, slim glass body with a bulb on the end. One of the two electrodes is called a reference electrode. It consists of a conductive wire submersed in a concentrated and stable salt solution. The other electrode is called the measuring electrode, which has a conductive wire in a solution of stable pH, surrounded by pH-sensitive glass. This glass allows a very small electrical current through it, with the voltage of the current being dependent on pH. The current travels through an opening—called a junction (very important)—in the reference electrode and creates a circuit between both wires. The voltage gets picked up by the connected system, then the system scales it according to known variables and outputs a pH measurement.
Please visit our other article about how pH electrodes work if you would like a more in-depth explanation.
Differential Electrode Design
Now, let’s move onto the differential electrode design.
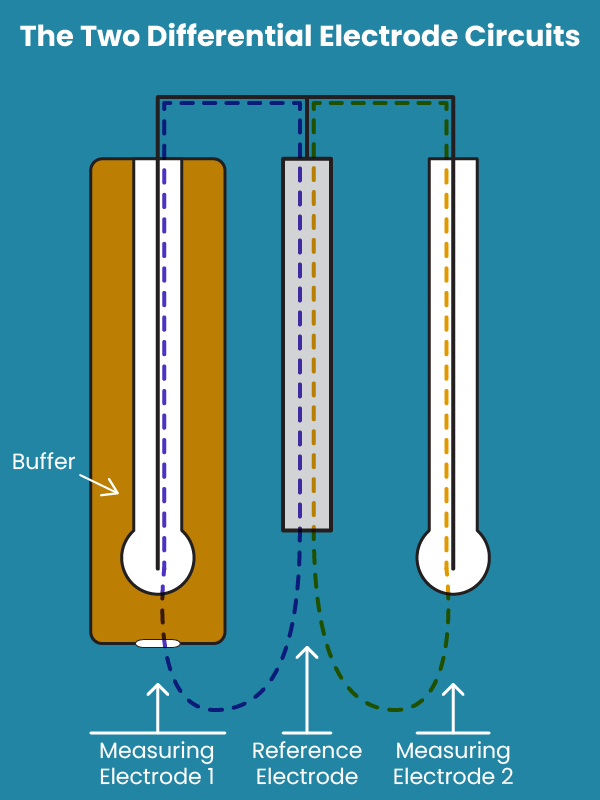
A differential electrode uses not two but three electrodes to perform its measurements. These consist of a reference electrode and two measuring electrodes. The measuring electrodes are very similar to those seen in standard pH sensors and are essentially a wire submersed in a solution and contained in pH-sensitive glass. Measuring Electrode 1 (ME1) is then submersed in a reference solution of known pH while Measuring Electrode 2 (ME2) is submersed in the sample.
The reference electrode is slightly different from the standard glass electrode. In a differential electrode, the reference electrode is a wire or conductive rod directly exposed to the sample. There is no salt solution or glass surrounding it.
There are actually two measurement circuits in a differential electrode: One is the voltage generated between the reference rod and Measuring Electrode 1 (in a solution of known pH). The second is the voltage generated between Measuring Electrode 2 (in the sample) and the reference rod. The difference between the voltage of the two measuring electrodes determines how far the sample is from 7 pH, hence the term “differential”.
Advantages of A Differential Electrode
A differential electrode has numerous benefits that result in better performance for longer periods of time.
Advantage 1 – Stability
A standard electrode has a reference solution that must be a salt solution with a particular ionic concentration around the wire for a stable voltage. If it gets diluted, then the measurement changes. In contrast, a more stable buffering solution that is not as affected by dilution may be used with differential electrodes.
Advantage 2 – No Electrode Poisoning
Standard reference electrodes have a wire that is exposed to the sample through the junction. When chemicals that react with the wire are present in the sample, then these chemicals change the voltage potential of the wire to negatively impact measurements. This is known as electrode poisoning.
When the differential reference electrode is attacked, it affects both circuits of the measuring electrodes (ME1 & ME 2) equally so the sensor output is not affected overall. See the equations below for mathematical examples of a chemical reaction with electrodes


Advantage 3 – Reduced Maintenance
Because the reference solution is more stable and the electrode can’t be poisoned, less maintenance is often required. In addition, electrodes need replacement less frequently, leading to significantly lower long-term maintenance costs.
Advantage 4 – Reduced Electrical Interference
As the reference electrode is common to both measuring electrodes, any electrical currents in the water are picked up by both circuits and subtracted. This removes the effect of many lower energy electrical currents on pH measurements, although some stronger currents may overload the electrodes. Some manufacturers claim that all ground loops are eliminated with the differential design, but it is still possible for some current to be conducted. The only way to completely remove them is with isolated inputs/outputs. That is why all pHionics sensors include electrical isolation.
Conclusion
Using a differential sensor comes with a number of benefits for many applications. If you are interested in purchasing one for a project, then the pHionics D-phi Series may be an excellent option. Our 4-20 mA differential pH/ORP Sensors are engineered to make the stability and ruggedness of differential electrodes available at a lower cost. They are also compatible with a wide range of systems and start at just $649. If you are worried about electrode poisoning or reference dilution in harsh industrial applications, then enjoy peace-of-mind and reduced expenses with the D-phi Series. Click on a parameter of interest below to learn more.
Please do not hesitate to reach out if you have any questions about water quality measurement or wish to speak with our sales team to see how we can help you.
Recent Articles

How Glass pH Electrodes Work
In this video, learn about the electrochemistry that allows silver/silver chloride pH electrodes to measure the acidity of solutions. Modern electrode design is also reviewed to demonstrate what improvements have been made and what weaknesses are still present. Click...
STs Series Sensor Storage
STs Series sensors are constructed from high-quality, durable materials that can be stored for long periods of time. The only weak point is the electrode, which can be damaged or expire during storage in the wrong conditions. These conditions vary depending on the type of electrode, which is why we have different storage instructions for each sensor.
How a Glass pH Electrode Works
A comprehensive article covering how glass electrodes measure pH in a simple, understandable format. Specifically for silver/silver chloride electrodes.
