Factors Affecting Dissolved Oxygen
Introduction
Regardless of what type of sensor is used to measure dissolved oxygen, there are three factors that must be accounted for to ensure accuracy. They are:
- Salinity
- Pressure
- Temperature
Each one influences the concentration at 100% saturation but temperature is unique because it also affects how oxygen interacts with sensor membranes. The rest of this article goes over how to account for these factors to ensure accurate results.
Note: Any reference in the article to maximum concentration or maximum amount of oxygen is a reference to 100% saturation and not supersaturation. Supersaturation refers to dissolved oxygen levels above 100% and occurs temporarily in places where water and oxygen are mixed extremely rapidly, such as waterfalls or hydroelectric plants.
For information on how modern dissolved oxygen sensors work, please see our article on Comparison of Dissolved Oxygen Sensors. That article compares the different methods and assists in determining which is best for your application.
How Salinity Affects Dissolved Oxygen
The definition of salinity is the concentration of salt dissolved in solution. When salt is introduced into water, it breaks up into ions with positive and negative charges that attract H2O molecules. This occurs because H2O is polar, meaning parts of the compound have slightly negative or positive charges. These are called partial charges and are expressed as δ, a lowercase delta, with + or – to indicate charge type. The partial charges attract water molecules to the oppositely charged ions from dissolved salts, as shown in the image below.
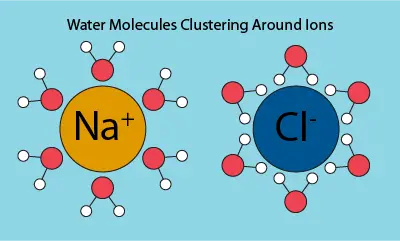
This grouping of ions and water molecules means there are fewer free water molecules for dissolved oxygen to bind to, allowing the gas to diffuse out of solution. As a result, increased salinity leads to reduced dissolved oxygen levels.
Salinity does not change how dissolved oxygen interacts with sensors, so changes in salinity do not affect concentration measurements (mg/L). Any sensors calibrated to measure percent saturation, however, must be properly adjusted to ensure accuracy.
As an example, let’s compare a zero salinity sample to seawater.
A sensor is initially calibrated to 100% saturation in water at 0 salinity, 760 mmHg, and 25°C is 8.26 mg/L. If that same sensor was placed in 100% oxygen-saturated sea water (34 salinity, 760 mmHg, 25°C), then it would read only 82% because the maximum saturation is 6.80 mg/L. A correction factor must be determined by measuring the salinity of the sample and cross referencing with a Salinity Correction Factor Table. This table is provided to the right. An example of how to calculate dissolved oxygen concentration with the correction factor is shown later in the article.
Dissolved oxygen sensors are commonly paired with conductivity sensors for the purpose of estimating salinity. The relationship between conductivity and salinity is defined by the Practical Salinity Scale of 1978, commonly abbreviated to PSS-78. While the scale is not linear or easily programmable into a monitoring system, pHionics has written an article explaining how salinity can be linearized for water quality monitoring systems such as Onset’s HOBO RX2100 or RX3000. A temperature-compensated conductivity sensor is required to linearize the scale. If you are in the market for one, we highly recommend the pHionics STs Series Conductivity for accurate measurements and long-term durability.
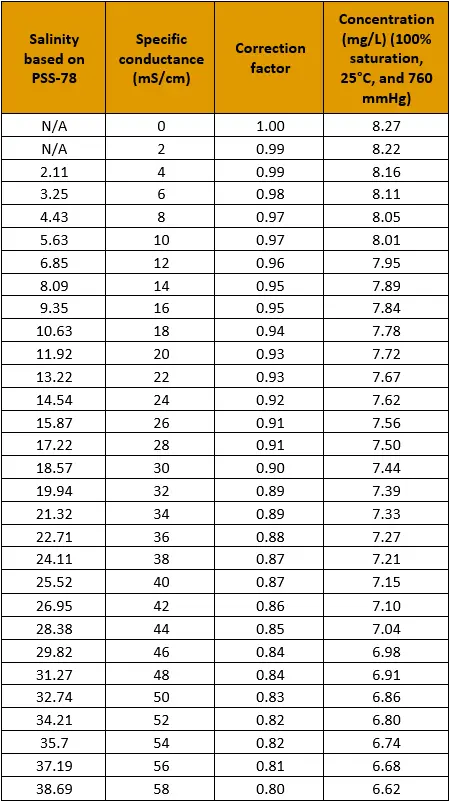
Table 1. Salinity and/or Conductivity Correction Factors for Dissolved Oxygen Measurements
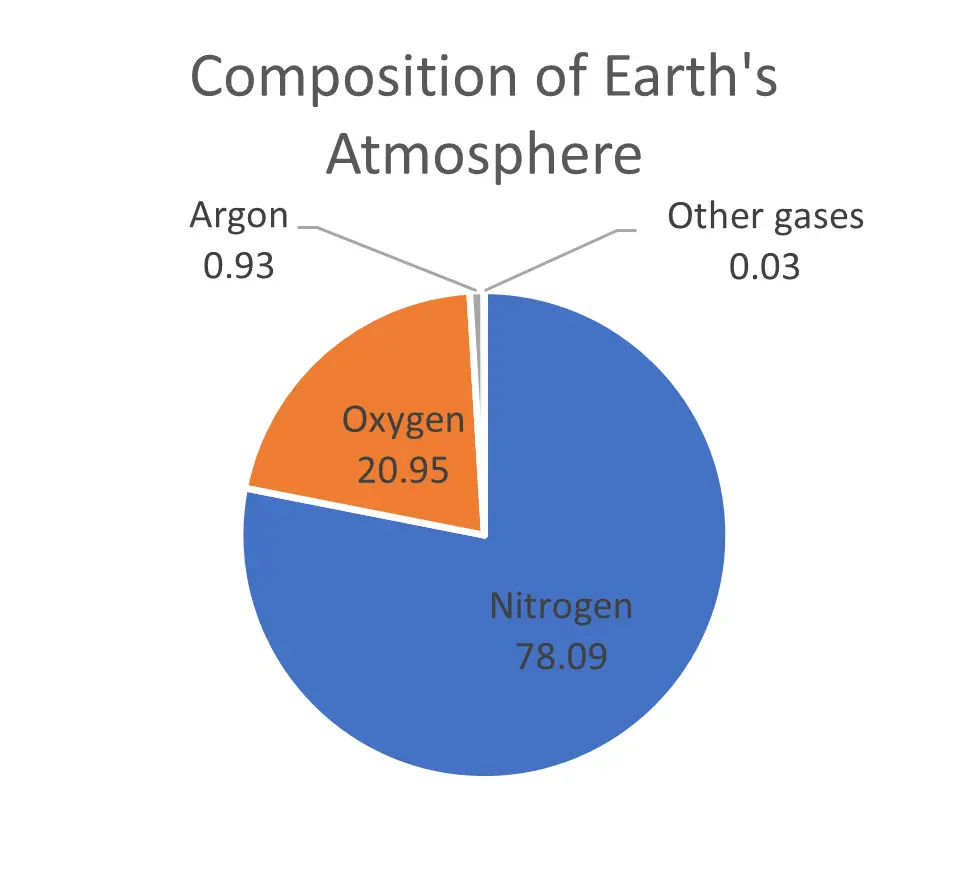
Oxygen makes up 21% of earth’s atmosphere, with the remaining percent being nitrogen and trace gases. As altitude increases, the percentage of oxygen does not change but the atmospheric pressure decreases. This means that the concentration of dissolved oxygen at 100% saturation decreases by the same percent that atmospheric pressure does, allowing us to calculate a barometric correction factor. An example is provided below:

Always ensure that the pressure being used is true barometric pressure, meaning it has not been corrected to sea level.
In summary, barometric pressure does affect maximum concentration but does not influence how dissolved oxygen interacts with sensors. Therefore, any instrument that is calibrated to measure concentration is not affected by changes in pressure but an instrument calibrated for percent saturation requires recalibration.
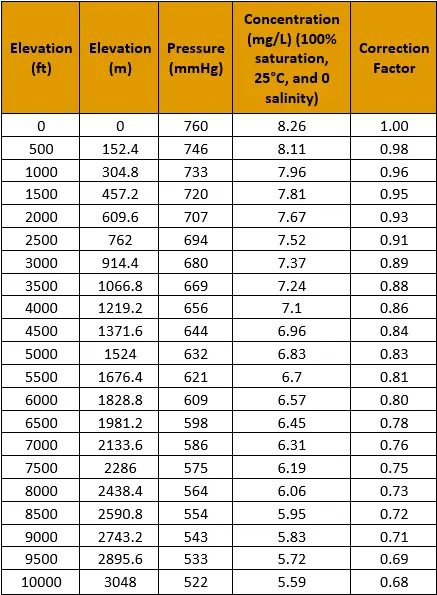
Table 2. Barometric Pressure Correction Factor Table
How Temperature Affects Dissolved Oxygen
Temperature and dissolved oxygen have an inverse relationship so, as temperature increases, dissolved oxygen decreases. This happens because an increase in temperature causes gas and water molecules to gain energy. As a result, the weak molecular interactions between water and oxygen gas are more easily broken, allowing oxygen to escape. Note that this relationship between temperature and saturation is true for all gases dissolved in solution.
Unlike salinity and pressure, temperature is the one variable that influences both percent saturation and concentration. Measurements are dependent on dissolved oxygen diffusing through the sensor membrane. As temperature increases, oxygen gas gains energy and more easily permeates the membrane, artificially increasing the sensor output. The inverse is seen at lower temperature. Most manufacturers, including pHionics, incorporate a thermistor in their instrument that automatically adjusts sensor output to account for changes in diffusion rate. This feature is called Automatic Temperature Compensation, which standardizes the output to 20 or 25°C depending on the manufacturer. This standardized temperature should be used when determining the correction factor for salinity and pressure as well.

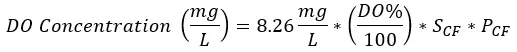
Example: Calculating what the dissolved oxygen concentration (mg/L) in seawater (34 salinity and 760 mmHg) is (using a sensor with Automatic Temperature Compensation to 25°C) if the sensor measures 84% saturation.
Conclusion
Hopefully, this article answered every one of your questions about variables that affect dissolved oxygen measurements. If not, then please reach out via our contact page and we will be happy to answer your question. The article will also be updated to include the answer and assist others who may have similar questions.
If you are in the market for a water quality sensor and would like a device that is durable, accurate, and hassle-free, we highly recommend looking into the pHionics STs Series™. All sensors are designed with high-quality materials, are isolated, and have long-lasting electrodes that are easily replaceable.
We wish you the best and hope you have a great day!
Additional Resources
Suggest an Article or Video Topic!
Recent Articles

How Glass pH Electrodes Work
In this video, learn about the electrochemistry that allows silver/silver chloride pH electrodes to measure the acidity of solutions. Modern electrode design is also reviewed to demonstrate what improvements have been made and what weaknesses are still present. Click...
STs Series Sensor Storage
STs Series sensors are constructed from high-quality, durable materials that can be stored for long periods of time. The only weak point is the electrode, which can be damaged or expire during storage in the wrong conditions. These conditions vary depending on the type of electrode, which is why we have different storage instructions for each sensor.
How a Glass pH Electrode Works
A comprehensive article covering how glass electrodes measure pH in a simple, understandable format. Specifically for silver/silver chloride electrodes.
