Comparison of Dissolved Oxygen Sensors
What is Dissolved Oxygen?
Dissolved oxygen is a measure of how many O2 molecules are present in a liquid (most often water) at any given time. It is incredibly important because oxygen sustains life in bodies of water and reduced dissolved oxygen levels can act as a warning for events that destroy ecosystems, such as algae blooms. The units of measurement are typically mg/L or on a scale of 0-100% saturation. The amount of dissolved oxygen that a liquid can contain is based on both chemical properties such as polarity and salinity, along with environmental conditions such as temperature and pressure.
Dissolved oxygen is introduced to water in three different ways:
Diffusion
- O2 molecules from the air naturally mix with water molecules when they interact at the surface of a body of water because there is typically a higher partial pressure of oxygen in air. Surface water, in general, is 100% saturation but dissolved oxygen levels fall as depth increases. Currents are responsible for bringing oxygenated surface water to organisms farther from the surface.
Photosynthesis
- Photosynthetic organisms release oxygen as a by-product of energy production, providing a large amount of aquatic life with dissolved oxygen. Photosynthesis is one method that allows dissolved oxygen to reach above 100% saturation, also called supersaturation.
Aeration
- Mixing of water and air, both naturally (e.g. waves) and artificially (e.g. hydroelectric dams), is another means of achieving supersaturation. These methods increase the surface area of a body of water, providing the opportunity for more diffusion to occur.
Dissolved oxygen leaves the water through two ways:
Aerobic respiration
- Many aquatic organisms require oxygen to survive and, after intaking it, exhale it as carbon dioxide molecules. A higher concentration of aerobic organisms in an area causes a drop in dissolved oxygen that can be lethal, which is why dissolved oxygen is so important to measure.
Diffusion
- Chemical concentrations always attempt to reach equilibrium, and oxygen is no exception. While diffusion can increase dissolved oxygen levels, it can also decrease dissolved oxygen levels when the partial pressure in water is greater than the surrounding atmosphere (e.g. when water is supersaturated).
Electrochemical Dissolved Oxygen Sensors
The original electrochemical (or amperometric) sensors were developed by Dr. Leland Clark in 1953 and the core technology has changed little since then. They have an anode and cathode surrounded by an electrolytic solution that is contained inside a plastic or glass body. A thin membrane on the tip of the electrode exposes the cathode to a sample. This membrane is permeable to oxygen (along with some other gases) and allows O2 to diffuse through at a rate proportional to the partial pressure of the gas in the sample. A difference in partial pressure is present because any oxygen that enters the electrode is reduced to OH– at the cathode. Reduction refers to when a compound gains electrons, and and example of the chemical reaction equation for commonly used metals is provided below:
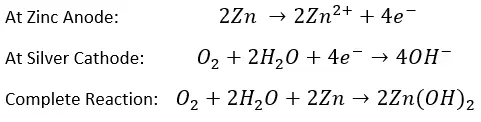
Redox equation of galvanic electrode with zinc anode and silver cathode
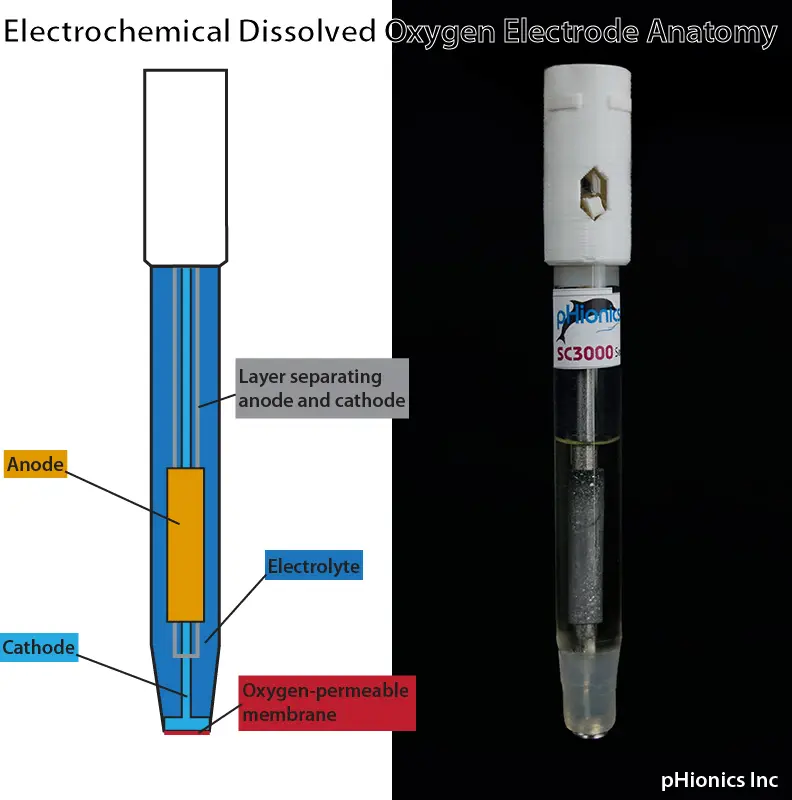
The acceptance of electrons by O2 causes a current–carried by the surrounding electrolyte–to flow between the anode and cathode. This current increases with a higher concentration of oxygen and the change is measured by the sensor.
The electrochemical method consumes oxygen, which poses an obstacle for measurement. While this does not affect the overall concentration of dissolved oxygen in the sample, it does affect the concentration in solution surrounding the tip of the electrode and causes artificially low readings. To overcome this, there must be constant mixing, or flow, of the sample. This means that the sensor is flow dependent and is important to be aware of when choosing what type of sensor is right for your application. This flow dependence is affected by the membrane materials and thickness, but those details are outside of the scope of this article. Manufacturers always state what the required flow rate is.
The electrochemical method is separated into two main types based on the metals used for the anode and cathode: galvanic and polarographic.
Galvanic Dissolved Oxygen Electrodes
Galvanic electrodes have anodes and cathodes with a large cell potential between them, meaning that one metal wants to give up electrons while the other wants to gain electrons. This potential is strong enough to cause electrons to flow from the anode to the cathode, in a process called self-polarization. In a sample with dissolved oxygen, the polarized cathode gives up electrons to any oxygen present and loses some polarization. As stated previously, electrons flow from the anode to replace ones lost to oxygen, resulting in current flow.
Oxidation of the anode results in formation of hydroxide precipitate, which eventually blocks the membrane and interferes with measurements. Be aware that, while presence of precipitate can affect performance, it does not automatically mean the electrode requires replacement. Many electrodes continue to function properly for months after visible build-up occurs.
Metals most commonly used in galvanic cells are lead or zinc for the anode and gold or platinum for the cathode.
Polarographic Dissolved Oxygen Electrodes
Polarographic electrodes have silver anodes and gold cathodes, resulting in a smaller cell potential that requires additional voltage to drive the transfer of electrons. Therefore, they are not self-polarizing like galvanic electrodes. This results in a warm-up time of 5-15 minutes before a polarographic electrode can be used.
Oxidation of the anode causes formation of silver chloride which coats the anode over time. Like the precipitate in galvanic electrodes, this coating eventually interferes with measurements. Because polarographic electrodes are not self-polarizing, this reaction does not occur when the electrode is not in use. As a result, polarographic electrodes can be stored for long periods of time without loss of life, whereas galvanic electrodes continue to be oxidized if exposed to oxygen.
The electrochemical electrodes described so far are known as steady-state electrodes, but there is one other variation worth mentioning. Polarographic electrodes can also be “pulsed”, meaning that a voltage is applied periodically instead of constantly. When the voltage is not being applied, dissolved oxygen from the sample can replace what was used up when the electrode was on. This means that pulsed polarographic measurements are not dependent on sample flow like steady-state electrodes are.
Galvanic vs. Polarographic Comparison

Optical Dissolved Oxygen Sensors (ODOs)
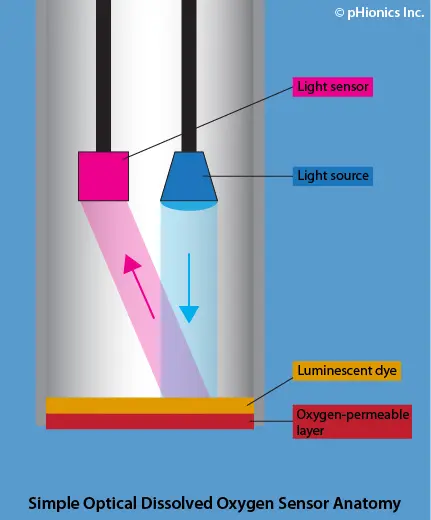
Optical (also known as luminescent) sensors utilize light to measure dissolved oxygen concentration. Like the electrochemical method, optical ODOs have an oxygen-permeable material at the tip of the electrode. An oxygen-sensitive, luminescent dye makes up the second layer. The dye is exposed to a specific wavelength of light in the sensor and the reflected light is picked up by a sensing element. The presence of oxygen causes changes in the lifetime and intensity of luminescence, which are quantified by the Stern-Volmer equation. This method does not use up oxygen so ODOs are not dependent on sample flow to maintain accuracy.
ODOs are separated into two types, based on whether they measure the change in lifetime or intensity of luminescence. The only difference between the two is their stability. Degradation of the dye layer over time is the main limiting factor of ODOs, and this break-down affects luminescent intensity more than lifetime. The dye layer is generally replaced once per year.
One significant difference between electrochemical and optical devices is their response time. On average, ODOs take twice as long to reach 95% accuracy compared to electrochemical (40 sec vs 20 sec, respectively), which is important to consider when measuring multiple samples.
Another major difference is their calibration frequency. ODOs maintain calibration well, often over months of time, whereas electrochemical devices generally only hold their calibration for a few days or weeks. Neither takes long to calibrate, and ODOs should still be checked for calibration consistently.
The last important distinction between the two dissolved oxygen measurement methods is their susceptibility to various compounds in a sample. Certain gases interact with electrochemical cathodes and interfere with measurements, while different chemicals attack the dye layers of optical sensors.
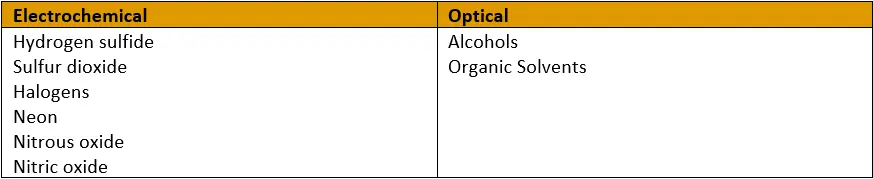
This list is from the YSI Dissolved Oxygen Handbook.
Conclusion
Each type of dissolved oxygen sensor has their pros and cons. To determine which type of sensor is best for your application, ask yourself the following questions:
How quickly does the sample flow? If there is no flow in the sample, then either pulsed polarographic or optical devices are needed.
How many samples are to be tested and how quickly does dissolved oxygen change in the sample? If there are many samples, or dissolved oxygen is changing rapidly, then electrochemical sensors are best.
How accessible is the sensor? If the sensor is hard to reach or in a remote area, then low maintenance ODOs may be best.
What chemicals are present in the solution? Electrochemical and optical sensors are affected by different compounds, as noted in the table above
For remote monitoring, what type of power source is being used? While optical sensors are more stable, they also use more power and may drain batteries faster than electrochemical. Longevity may be more important than drift and require an electrochemical sensor.
If you are interested in galvanic sensors, pHionics produces the STs Series Dissolved Oxygen. It includes a dissolved oxygen sensor with an isolated, 2-wire, 4-20 mA transmitter in one device. An anode with a large surface area surrounded by a large volume of electrolyte allows our electrodes to last an average of two years in typical applications before requiring replacement. Combination with a 2-wire transmitter provides extremely simple set-up and high accuracy signal transmission. For more information, please see our product page.
Suggest an Article or Video Topic!
Recent Articles

How Glass pH Electrodes Work
In this video, learn about the electrochemistry that allows silver/silver chloride pH electrodes to measure the acidity of solutions. Modern electrode design is also reviewed to demonstrate what improvements have been made and what weaknesses are still present. Click...
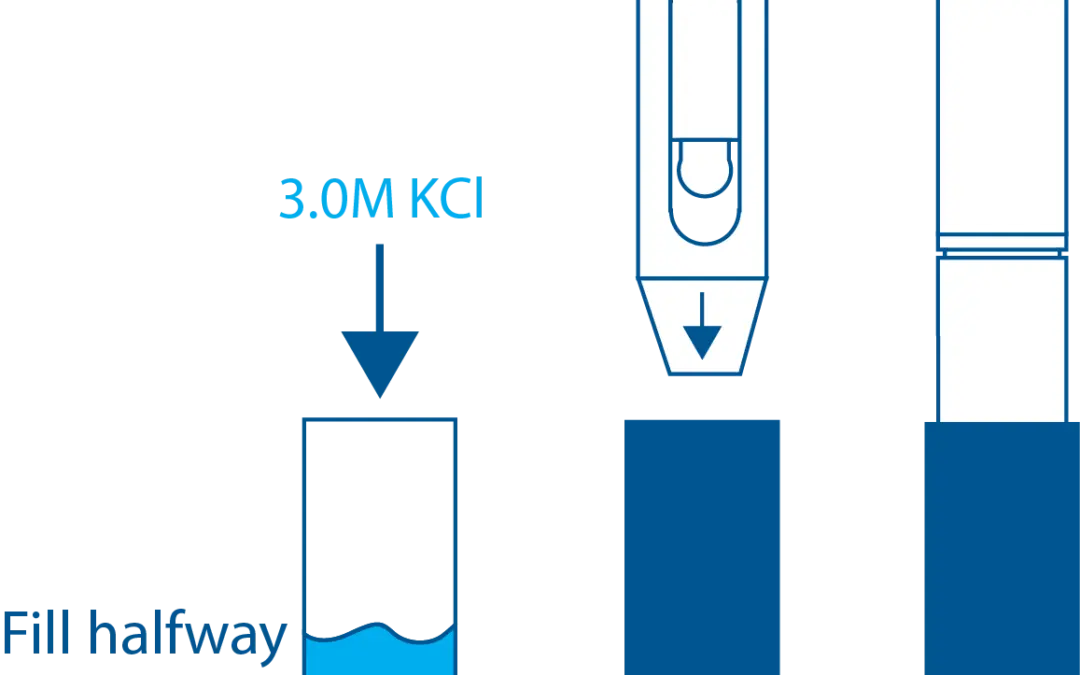
STs Series Sensor Storage
STs Series sensors are constructed from high-quality, durable materials that can be stored for long periods of time. The only weak point is the electrode, which can be damaged or expire during storage in the wrong conditions. These conditions vary depending on the type of electrode, which is why we have different storage instructions for each sensor.
How a Glass pH Electrode Works
A comprehensive article covering how glass electrodes measure pH in a simple, understandable format. Specifically for silver/silver chloride electrodes.
