The Basics of Oxidation-Reduction Potential (ORP)
Introduction
Oxidation-reduction potential, or ORP for short, is the measure of how likely a solution is to give or receive electrons given the right circumstances. Some common applications involve pollution control and as a measurement of biological activity such as fermentation. ORP can be difficult to interpret, however, for reasons that will be discussed later in this article. To start, we define what an oxidation-reduction reaction is, then dive into how ORP is calculated and how sensors measure it.
What are Oxidation-Reduction Reactions?
Oxidation-reduction (redox) reactions are defined as the process of electrons being transferred from one atom or ion to another. The molecule that accepts electrons is reduced because it now has a lower charge, while the molecule that donates electrons is oxidized. As electrons cannot exist independently in solution, a reduction cannot occur without oxidation and vice versa.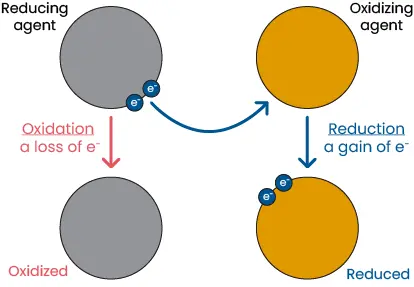
A complete redox reaction is shown below.
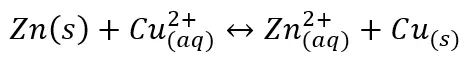
Redox reactions are often broken up into half reactions. Each half reaction has a standard reduction potential expressed in volts or millivolts, with more positive values indicating how spontaneous the reaction is, or how likely the product is to be formed given the right conditions. One of the most common examples in chemistry classes is the redox reaction of solid zinc in a copper solution. When zinc is placed into a copper solution, the zinc is slowly plated with solid copper over time. The reason for this becomes clear when we look at the standard reduction potentials (E°) and calculate the oxidation-reduction potential (ORP).
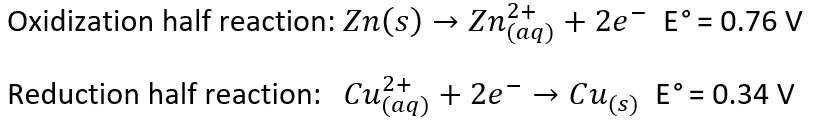
The sum of the half reaction potentials, 0.76 and 0.34 V, gives an ORP of 1.10 V in standard conditions. Because 1.10 V is positive, this indicates that the reaction is favorable and occurs spontaneously. Higher voltages indicate that the reaction is more likely to occur, while lower or negative voltages are less likely to occur.
Again, this is a measure of the potential for the reaction to occur and does not indicate that the reaction happens in any conditions. The reaction only occurs given proper conditions such as sufficient activation energy.
Standardizing Redox Reaction Potentials
Standard reduction potentials are “standard” because the given values are in relation to the voltage of hydrogen reduction, which has a potential of 0 V with the assumed conditions:
- 25°C
- 1M concentration of aqueous species
- 1 atm partial pressure
Standard hydrogen reduction reaction
As real-world measurements always have conditions differing from standard, actual reaction potentials vary. An equation developed by Walther Nernst, called the Nernst Equation, accounts for differences in temperature and concentration from the assumed conditions. The equation is provided below:
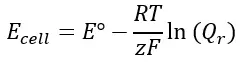
- Ecell = the ORP value at the temperature and concentration of interest
- E° = the standard potential
- R = the universal gas constant (8.314 J/(Kmol))
- T = the temperature in Kelvin
- z = the number of electrons being transferred
- F = Faraday’s Constant (96,500 C/mol)
- Q = the quotient of the reductant and oxidant
Example: Calculate the ORP of the reaction between copper and zinc when [Cu2+] = 1.5 M and [Zn2+] = 0.5 M. E° = 1.10 V
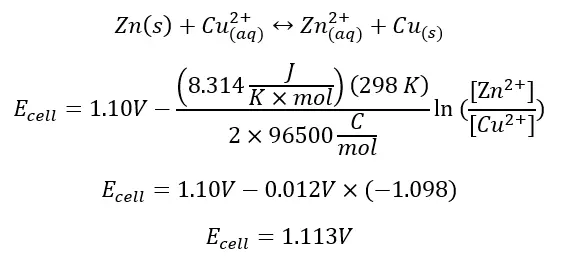
Remember that the standard potential E° is calculated with the assumption that there is 1 M of the reactant and product. Because there is more reactant than product, there is higher reaction potential than the standard E°, indicating the reaction is more favorable in these conditions.
What is ORP?
In terms of water quality, ORP is a measure of all reaction potentials present in the sample solution. Therefore, ORP is considered non-specific because it does not measure any unique species, unlike pH that specifically measures hydrogen ion concentration.
ORP is expressed in volts or millivolts and typically ranges from -1000 to +1000 mV. A positive voltage indicates that the solution is more likely to oxidize, or take electrons, from a species (compound, atom, or ion) with lower oxidative potential. For example, a +1000 mV solution can oxidize a +800 mV solution and a +800 mV solution can oxidize a +100 mV solution. The opposite is true for a negative voltage.
Applications
By measuring ORP and using the Nernst Equation, it is theoretically possible to determine the concentrations of redox reactants and products in solution. Unfortunately, this can be quite difficult to do because ORP measurements are non-specific. In addition, ORP is heavily influenced by pH. As a result, ORP measurements are not useful in comparing two different water sources. Instead, ORP is best used to compare changes in a single solution over time. Changes may indicate the following:
- An increase in pollution such as heavy metals
- Bacterial activity (fermentation, nitrification, etc.)
- Redox reaction completion (Cyanide or chromate reduction)
How ORP is Measured
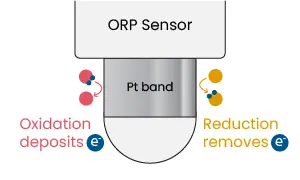
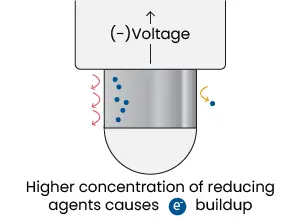
ORP measurements are performed with electrochemical sensors made of two electrodes. One is a reference electrode which, just like a pH sensor, uses a silver/silver chloride wire with KCl reference solution. This provides a stable point to compare the voltage that develops on the second electrode. The second electrode is called a measuring electrode, which has a band or coating made of inert metal (platinum or gold) that is exposed to solution. Platinum is commonly used because the element acts as a catalyst to redox reactions and freely gives or accepts electrons, thereby allowing a potential to build up that matches the solution. In addition, the metal also resists corrosion to provide stable long-term measurements of ±5 mV.
Please note that the two electrodes are normally put together into a single combination electrode.
Conclusion
Hopefully, this article provided an effective look at the basics of ORP measurement. While somewhat more difficult to utilize than specific measurements such as pH, ORP still finds many uses in industrial and environmental monitoring.
For any questions or feedback, please click on the Contact Us button below. We would love to hear from you!
If you need to measure ORP for any environmental applications, then we highly recommend the pHionics STs Series™ ORP sensor. A slim design (0.75” dia.) allows the STs Series to go where others can’t while providing highly accurate measurements using proprietary noise-reduction technology.
Suggest an Article or Video Topic!
Recent Articles

How Glass pH Electrodes Work
In this video, learn about the electrochemistry that allows silver/silver chloride pH electrodes to measure the acidity of solutions. Modern electrode design is also reviewed to demonstrate what improvements have been made and what weaknesses are still present. Click...
STs Series Sensor Storage
STs Series sensors are constructed from high-quality, durable materials that can be stored for long periods of time. The only weak point is the electrode, which can be damaged or expire during storage in the wrong conditions. These conditions vary depending on the type of electrode, which is why we have different storage instructions for each sensor.
How a Glass pH Electrode Works
A comprehensive article covering how glass electrodes measure pH in a simple, understandable format. Specifically for silver/silver chloride electrodes.
