Managing Water Quality in Aquaculture
Introduction
Annual demand of seafood has increased significantly over the past 50 years, from less than 50 million metric tons to over 175 million metric tons. Due to the unsustainable nature of fisheries catching wild seafood, the aquaculture industry has accounted for most of this increase. This growth is expected to continue into the future to match the world’s population growth.
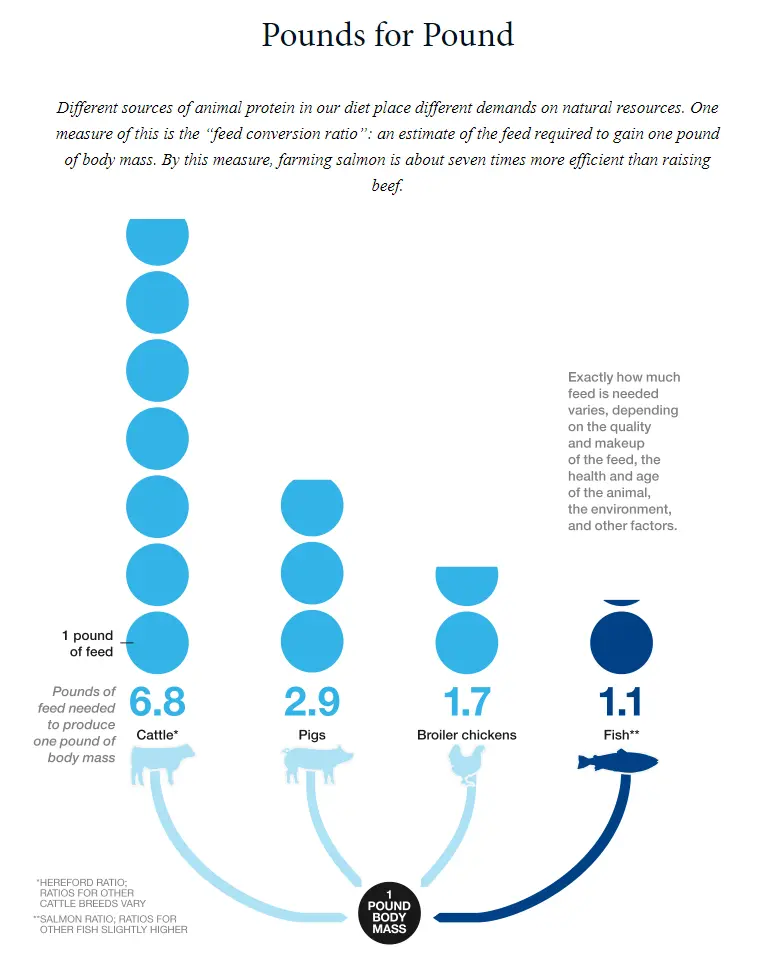
Aquaculture is found to be one of the most efficient means of meat production, as shown by the feed conversion ratio (picture below) provided by National Geographic in the article How to Farm a Better Fish. In many cases, however, farmed seafood is not nearly as efficient as it could be. This is due to poor water quality inhibiting livestock growth.
Exact methods of improving conditions vary depending on the type of aquaculture setup, so this article focuses on what parameters must be controlled and how to measure them, as opposed to detailed methods of control.
Methods of Measurement
Before diving into specific water parameters, let’s review different measurement techniques for them. These include colorimetry, benchtop meters, handheld meters, and mounted sensors.
Colorimetric
This method involves obtaining a water sample and adding specific chemicals that change color depending on the concentration of a parameter (e.g. ammonia levels). These tests are evaluated either by eye in the field or by spectrophotometry in the lab. Colorimetry is generally low-cost (if evaluated by eye) and requires little technical experience to understand. It is also the least precise and is slower than most other methods.
Benchtop meter
Benchtop meters use electrochemical devices to precisely determine a given water quality parameter. They are used when water samples are collected periodically (once per day or week) and brought to a central lab so numerous water quality parameters can be tested on the same sample. Different meter models range in accuracy, speed, and cost, but are generally more accurate, faster, and more expensive than colorimetric tests.
Handheld meter
Both benchtop and handheld meters are used for periodic sampling but handheld meters have the benefit of testing one or more points in your water source in real-time. Most parameters are stratified in the water to some degree so measuring multiple points in one visit helps obtain a clearer understanding of water conditions. A handheld meter generally is similar speed, lower accuracy, and lower cost than a benchtop meter. The increased portability makes it a good option for testing multiple ponds or tanks.
Mounted sensor
Mounted sensors are used for continuous real-time monitoring of key parameters whose rapid change may cause livestock loss. They often connect to alarm or control systems that alert an operator if parameters are approaching dangerous levels. In a more complex setup, sensors automatically trigger pumps, filters, or aeration and only require human intervention if errors occur.
Mounted sensors provide the most complete and accurate monitoring for safety of livestock but are the most expensive. As such, they are mainly used for life-or-death parameters like dissolved oxygen. Automating measurements and corrective actions wth sensors reduces cost in the long term because fewer employees are required for operation and resources are more used more efficiently.
Important Water Quality Parameters for Aquaculture
Ammonia
Compounds containing nitrogen are added to aquaculture systems through food (especially high-protein food) and fertilizer. Much of the introduced nitrogen is broken down by livestock, bacteria and plants to produce ammonia.
Ammonia is found in two forms—one form that is toxic and one form that is significantly less toxic.
- The toxic form is known as un-ionized ammonia (NH3) and reduces livestock health at levels above 0.02 mg/L.
- The significantly less toxic form is called ammonium (NH4+). There is almost always some amount of ammonia present, but which form it takes varies greatly depending on the temperature and pH.
At 7 pH or below, nearly all the ammonia (approx. 99%) is in an ionized form. Above 8 pH, the toxic form becomes present at anywhere from 1-10% of the total ammonia concentration. The actual percentage depends on temperature, with higher temperatures causing an increase in un-ionized ammonia. A table is provided below with exact percentages.
Percentage of Un-Ionized Ammonia at Specific pH and Temperature
While keeping ammonia in a less toxic state prevents deaths, it is best to avoid build-up entirely. Ammonia removal is done with a strong filtration system, nitrifying bacteria, fast-growing plants, or a combination of the three.
Monitoring Ammonia in Aquaculture
With neutral pH, good filtration, and healthy nitrifying bacteria, it is relatively easy to control ammonia. As such, periodic measurements (weekly-biweekly) via any of the previously discussed methods are acceptable.
Ammonia should always be tested for when livestock experience any illness. Increased testing frequency may be required after ammonia spikes occur.
For more information, please visit the following sources:
Temperature
Temperature is one factor that significantly affects the growth rate of farmed fish. Higher temperatures increase their metabolic rate while lower temperatures do the opposite. Each species differs in their preferred temperature ranges so staying up-to-date on the latest research is important for an efficient operation.
While temperature is important for the health of the fish, the ability to control this parameter depends on the type of aquaculture system used. Any operations built inside or exposed to natural waterways have trouble controlling this factor because heating an entire stream or lake is both cost-prohibitive and extremely detrimental for the surrounding environment.
Inland pond cultures or indoor operations have a much easier time monitoring and controlling temperature because their systems are typically smaller. Small systems are also more sensitive to temperature fluctuations, making monitoring more important.
Monitoring Temperature in Aquaculture
Temperature may be measured by any handheld meter or mounted sensor, depending on the desired measurement frequency. Temperature measurements are often included with other water quality instruments so a discrete sensor may not be required.
pH
pH is the number of free hydrogen ions (H+) in an aqueous solution. It is measured on a logarithmic scale from 0-14 pH. A solution is more acidic (< 7 pH) when more H+ ions are available, and more basic (> 7 pH) when less are available. The optimal range for most aquatic organisms is between 6-8 pH, depending on the species.
Nearly all aquaculture systems experience a pH level that cycles throughout the day due to photosynthetic organisms. Photosynthesis converts acidic CO2 into O2 when exposed to sun, slowly causing an increase in pH throughout the day. At night, the opposite occurs.
As discussed in the Ammonia section, higher pH conditions create a more toxic environment due to an increase in un-ionized ammonia. Knowing when these conditions occur is imperative to the survival of livestock.
Why pH Cycles Throughout the Day
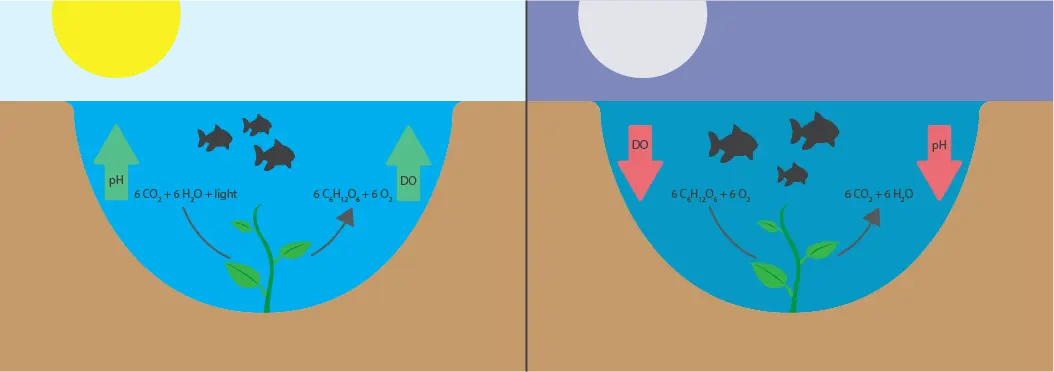
During the day, sunlight reaches plants and other photosynthetic organisms living in water. They perform photosynthesis, which removes acidic carbon dioxide (CO2) to produce dissolved oxygen (DO) and glucose (C6H12O6). When sunlight is not available (at night or from cloudy weather), photosynthetic organisms switch to respiration to use the energy stored in glucose, breaking it down to release CO2 and decrease pH. pH and DO are typically lowest right before sunrise.
Monitoring pH in Aquaculture
The pH balance is fragile and may quickly exceed healthy levels for fish if not properly maintained, especially in smaller systems. As such, it is one water quality parameter that should be measured constantly with a mounted sensor to ensure preventative measures are taken before any fish die off occurs. A fully automated system that maintains the pH balance significantly reduces manual labor and improves the health of fish in the long run.
Alkalinity
Alkalinity (AKA KH) is the buffering capacity of water, meaning how resistant water is to changes in pH. It is typically expressed as ppm or mg/L calcium carbonate and is a measure of how many negative ions (Carbonate and bicarbonate) are present. These compounds bind with excess hydrogen ions that would otherwise turn the water more acidic, thereby limiting changes in pH until the alkaline ions are used up. Ideal ranges are between 50 and 200 ppm.
Alkalinity values depend on the surrounding geology, as different soils/stones–such as limestone–release more alkalinity than others. In systems where alkalinity is low, it is often necessary to adjust levels by adding limestone or sodium bicarbonate to prevent changes in pH. Weather and other seasonal variations also change alkalinity concentrations by evaporating or adding water through rainfall.
Nitrification–the conversion of ammonia into less toxic compounds–is one process that significantly reduces levels of alkalinity in a matter of days because it releases hydrogen ions. This is one reason why controlling the ammonia levels in aquaculture systems is essential.
Monitoring Alkalinity in Aquaculture
See Monitoring Hardness.
Hardness
Hardness, or Total Hardness, is the opposite of alkalinity. It is a measure of positive divalent ions (calcium, magnesium, strontium, iron, and manganese) present in water. It is, somewhat confusingly, also measured in ppm or mg/L calcium carbonate and should be above 50 ppm. Hardness and alkalinity often track each other because salts used to change one parameter, such as calcite (CaCO3), usually have a cation (Ca2+) and an anion (CO32-) that increase both. Levels change depending on the surrounding environment, as some soils leach ions into the water while others remove them.
Monitoring Hardness in Aquaculture
Measuring both alkalinity and hardness involve the use of colorimetry and a meter (typically benchtop). A titration is performed on a water sample by adding acid or base until the sample reaches a certain pH, at which point a color change occurs. The amount of acid or base added is directly proportional to alkalinity and hardness. These tests should be performed periodically (weekly to monthly) depending on how much they change. Continuous monitoring of pH also provides an estimate buffering capacity in the water based on pH fluctuations, further reducing the need for testing alkalinity and hardness. Alkalinity and hardness analyzer systems are available that perform these tests automatically, but they are not generally required for aquaculture due to the infrequency of testing.
Check out this review for an in-depth look at alkalinity and hardness:
Alkalinity and Hardness: Critical but Elusive Concepts in Aquaculture
Salinity
Salinity is a measure of the amount of salt dissolved in water and is typically expressed in ppt or mg/L, though the official salinity scale in research is technically unitless. This water quality can change when a significant amount of water leaves (e.g. precipitation) or enters (e.g. rain) the system so salinity varies with weather and season. Ideal ranges depend entirely on the species.
Monitoring Salinity in Aquaculture
In most aquaculture systems, salinity varies only slightly day-to-day, so infrequent measurement (weekly-monthly) is required using a refractometer or salinometer. For systems that experience significant, rapid changes which may be detrimental to livestock, continuous monitoring using a conductivity sensor is recommended for preventative measures to be taken.
Dissolved Oxygen
Dissolved oxygen is a measurement of how much O2 is present in a liquid at a given time. It is measured in mg/L or percent saturation and healthy ranges vary significantly between species. Dissolved oxygen is the most important water quality parameter to monitor because oxygen levels can drop extremely quickly, resulting in fishkills after a few hours. Even slightly reduced levels induce stress and increase susceptibility to disease, leading to lower growth rates or the loss of livestock. To prevent this, consistent monitoring of dissolved oxygen is necessary, along with a good aeration system to counteract when levels decrease. Understanding how and when dissolved oxygen levels drop is important to properly time the aeration system and avoid wasting resources by running aeration systems longer than necessary.
Temperature, pressure, and salinity all affect the amount of dissolved oxygen that water can hold. While pressure and salinity are not likely to change rapidly, knowing the lows and highs of temperature throughout the day allow for preventative actions to be taken. Check out our article Factors Affecting Dissolved Oxygen for more information on the mechanisms behind these three environmental factors.
Other factors that influence dissolved oxygen include livestock oxygen use, amount of photosynthetic biomass, and weather.
-
- As livestock grow, so do their dissolved oxygen requirements. Knowing their total biomass is key to understanding exactly how much they need and how quickly the current supply could be used up.
- The amount of photosynthetic biomass is also important to be aware of because, while these sources produce oxygen during the day, they use it up at night during respiration. Fishkills occur most often before sunrise because that is when oxygen levels are lowest.
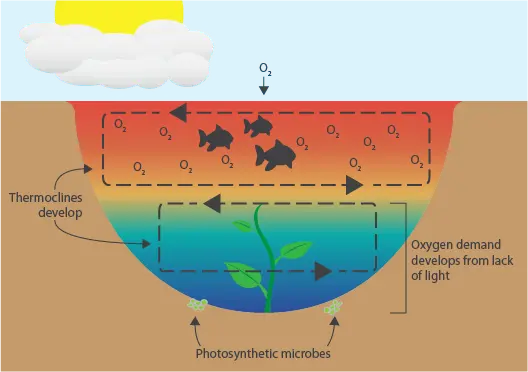
- Specific weather conditions also affect dissolved oxygen levels. When the weather is cloudy, little sunlight reaches photosynthetic organisms in the water and they switch to respiration. If it is also a warm day, then a thermocline develops that stratifies the water, circulating the oxygenated water at the top while depriving the depths of oxygen. Organisms at oxygen-deficient depths develop oxygen demand that cannot be met. Warm, cloudy days are common before a storm in the summer so, after this oxygen demand develops, wind and rain follow to disrupt the thermocline that develops. While this may sound like a good thing, oxygen-deprived organisms quickly intake the oxygen when it reaches them, causing a severe drop in oxygen that may lead to a fishkill. See the set of images to the right (desktop) or below (mobile) for a visual example of this phenomenon.
Signs that oxygen is depleted include:
-
- Fish near surface gulping for air (piping)
- Fish stop feeding
- Rapid change in water color due to algal bloom
- Strong stench from water
- Extended period of hot, cloudy weather
- Heavy summer wind and rainstorm
How Weather Causes Low Dissolved Oxygen Levels
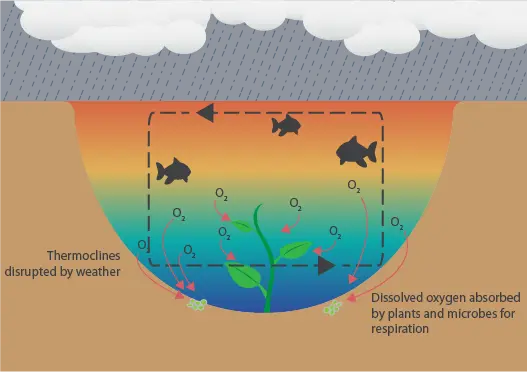

Monitoring Dissolved Oxygen in Aquaculture
Because dissolved oxygen changes rapidly and is extremely important to livestock, dissolved oxygen should be monitored frequently (continuously – twice per day) using either a handheld meter or mounted sensors. The safest method of measurement is using one or two mounted dissolved oxygen sensors placed in a spot with the highest and/or lowest levels to get a clear idea of dissolved oxygen stratification.
Intelligent Aquaculture
Technology is evolving rapidly, allowing the world of aquaculture to experience the benefit of everything being connected through the IoT. This starts with the use of sensors, cameras, and other data acquisition devices to monitor the various parameters mentioned in this article. The gathered information is then sent to a control center where it is processed and stored. After that, a computer uses the data to determine if any actions must be taken to optimize the aquaculture system, whether in filtration, feed weight, aeration, etc., based on historical values. Nearly every process can be controlled remotely with just a few skilled overseers. This IoT method of aquaculture can improve productivity, quality, and sustainability of the operation while reducing expenses.
There are certainly issues with implementing new technology into the process that must be addressed. While there may be long-term savings from installing and using new equipment, the initial costs are quite high, especially for smaller operations. There is also an issue with the robustness of these systems, in terms of their ability to handle equipment failures, power loss, or network connectivity issues. It also may be difficult to upgrade or incorporate new technologies once one system is in place.
Please visit this article for more information on the topic:
Transitioning Toward the Future of Aquaculture
Because the technology is still developing and expensive, it is important to consider the best method of transitioning into intelligent aquaculture for your particular situation. Continuous monitoring of key parameters such as dissolved oxygen and pH may be worth investing in since that can prevent a sudden, disastrous loss of livestock. A pump controlled by a PLC (programmable logic controller) that automates pH management might be the next technological addition. Obviously, the exact steps vary depending on your operation, but the start is data collection with sensors.
Just as your personal information is valuable to big tech companies to predict your behavior, knowing the exact parameters of your operation at any point in time is valuable to protect your livestock. It also assists in determining what technology is required in the future. Choosing sensors that are accurate, robust, and compatible with future technologies is the first step.
If you are interested in moving toward the future with intelligent aquaculture, we highly recommend the pHionics STs Series–highly accurate, easy-to-use, and durable sensors designed for any system that offers analog inputs. Please visit our products page to learn more about our pH, dissolved oxygen, or conductivity sensors.
Conclusion
With the information outlined in this article, it should be easier to determine how and when to measure the many parameters that influence aquaculture success. Dissolved oxygen and pH are the most important due to their variability so should be monitored consistently. pH also provides information on toxicity of nitrogenous waste, alkalinity, and hardness, so these parameters do not require as frequent measurement. Salinity may rarely require testing. Through proper monitoring of these parameters, it is possible to reduce labor costs and improve efficiency of an operation, allowing aquaculture to reach it’s potential to feed the world.
If you have any questions or concerns about this article or would like to suggest a topic for an upcoming one, please reach out using the form below.
Thank you for reading and have a great day!
Suggest an Article or Video Topic!
Recent Articles

How Glass pH Electrodes Work
In this video, learn about the electrochemistry that allows silver/silver chloride pH electrodes to measure the acidity of solutions. Modern electrode design is also reviewed to demonstrate what improvements have been made and what weaknesses are still present. Click...
STs Series Sensor Storage
STs Series sensors are constructed from high-quality, durable materials that can be stored for long periods of time. The only weak point is the electrode, which can be damaged or expire during storage in the wrong conditions. These conditions vary depending on the type of electrode, which is why we have different storage instructions for each sensor.
How a Glass pH Electrode Works
A comprehensive article covering how glass electrodes measure pH in a simple, understandable format. Specifically for silver/silver chloride electrodes.
