D-phi Series™ Features
Simplified explanation
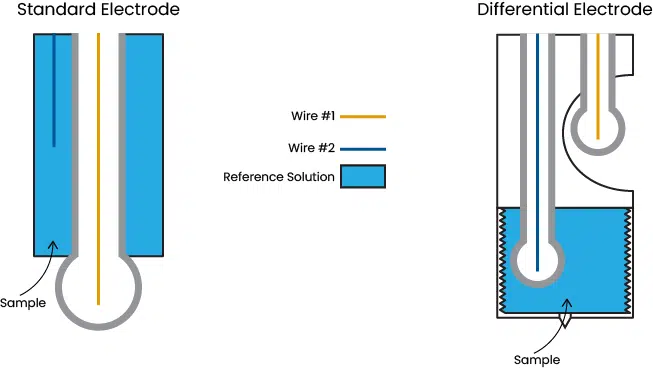
A typical pH/ORP electrode compares the voltage between two wires, one of which is enclosed in a glass bulb while the other is submersed in a reference solution that contacts the sample. Contacting the sample is key to a responsive system, but it also introduces two weaknesses.
- The salt solution surrounding Wire 2 gets diluted by the sample over time.
- Wire 2 may be coated or react with chemicals in the sample, affecting response time and accuracy. This is known as electrode poisoning.
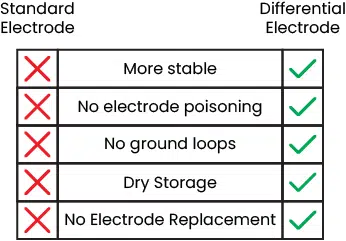
Differential electrodes eliminate these problems by encasing both wires in glass bulbs. The sample also cannot contact either wire so the electrode cannot be poisoned.
One of the glass bulbs is exposed to the sample, while the other is surrounded by a reference solution. For differential electrodes, a strong buffer may be used instead of a salt solution. The differential reference solution does still experience dilution but, by the nature of a buffer, it stays more stable than the salt solution used in typical pH electrodes. When dilution eventually affects the differential electrode solution, it is easily replaced within a minute using the reference capsules included with each sensor. Additional reference capsules are available for purchase.
Oh, and differential electrodes may be stored dry without adverse effects on performance.
Isolated
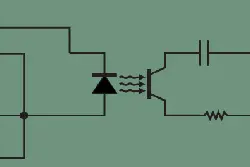
Having an isolated sensor prevents ground loops, which improves accuracy and prevents electrocution of the user. Isolation does this by preventing current flow from one system to another while still allowing information or energy exchange. This is accomplished using optocouplers and transformers inside D-phi Series sensors. If you are interested in learning more, then please read our article about isolation and its benefits in water quality measurement.
A circuit diagram including an optocoupler, which is a common method of electrical isolation.
Preamplifier
Most sensors on the market directly send their measurements to a receiver without modification. These measurement signals are small voltages that are vulnerable to electromagnetic interference. D-phi Series sensors include a transmitter inside with a built-in preamplifier, which strengthens signals to reduce interference picked up along the wires.
For more information on preamplifiers or differential amplification, please read our article on Eliminating Electromagnetic Interference.
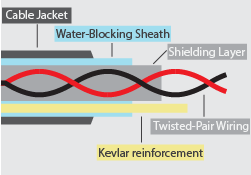
Heavy Duty Cable
pHionics always uses high quality materials designed to provide many years of service, and our cable is no exception. Kevlar reinforcement prevents stretching to maintain proper depth of measurement. Kevlar also reduces the chance of the cable being cut through, thereby preventing loss of the sensor. In addition, water-blocking technology prevents liquid from entering a cut cable and traveling into attached electronics. Finally, chemical resistance is provided by a polyurethane coating and electrical interference is prevented with shielding.
Reversible Input Protection and Operation
Improper wiring is an easy way to waste hours of time troubleshooting. It also has the potential to damage your expensive sensors if current is applied along the wrong wire.
Reversible input protection is a feature that prevents both problems to create a water quality sensor that is easy to install without worries of improper wiring that may damage equipment. The two wires for each channel are interchangeable to power the sensor or output the 4-20 mA signal, as shown in the diagram below.
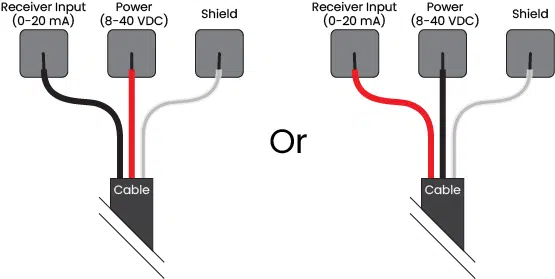
Or enhance your knowledge through our
Featured Article
The Guide to pH Drift
pH drift is one of the most frustrating issues to troubleshoot because it can seem random and be caused by many different aspects of the environment, maintenance, and sensor design. This guide provides steps to simplify the troubleshooting process and determine what the cause of drift is along with how to fix it.