How Temperature Affects pH Measurements
Table of Contents
Introduction
Understanding factors affecting the accuracy of a measurement is essential to get the most value out of water quality sensors. Once understood, then each of them can be accounted for by various means, either by the user or the sensor itself.
For pH measurements, temperature plays a large factor and can contribute to significant error if not understood and compensated for. The rest of this article covers how temperature affects pH sensors and the true pH of the sample, along with how to correct for these temperature effects.
How Temperature Affects pH Sensors
First, we need to give a little background on how pH sensors work. pH measurement is reliant on developing an electrical potential between a reference and sample solution. This potential is very small—in the millivolts range, to be exact—so any changes in material structure can have a noticeable effect. As a result, temperature affects the following components:- The reference and measuring elements have increased resistance at higher temperatures and vice versa for lower temperatures.
- The glass bulb also experiences the same change in resistance with temperature, causing a difference in potential energy that builds up.
- The junction potential that builds up at the reference junction changes with temperature.
- Solutions contained in the measuring and reference chambers have ionic compounds that vary in solubility with temperature. As such, conductivity of the solutions change with temperature.
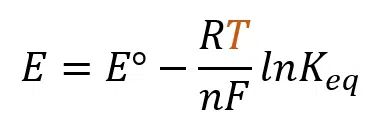
Image 1. Temperature (T) variable highlighted in the Nernst equation.
This equation allows one to calculate the electrode potential and slope at a given temperature, which is all the knowledge needed for this article. Images 2 and 3 provide examples of how electrode potential changes with temperature:
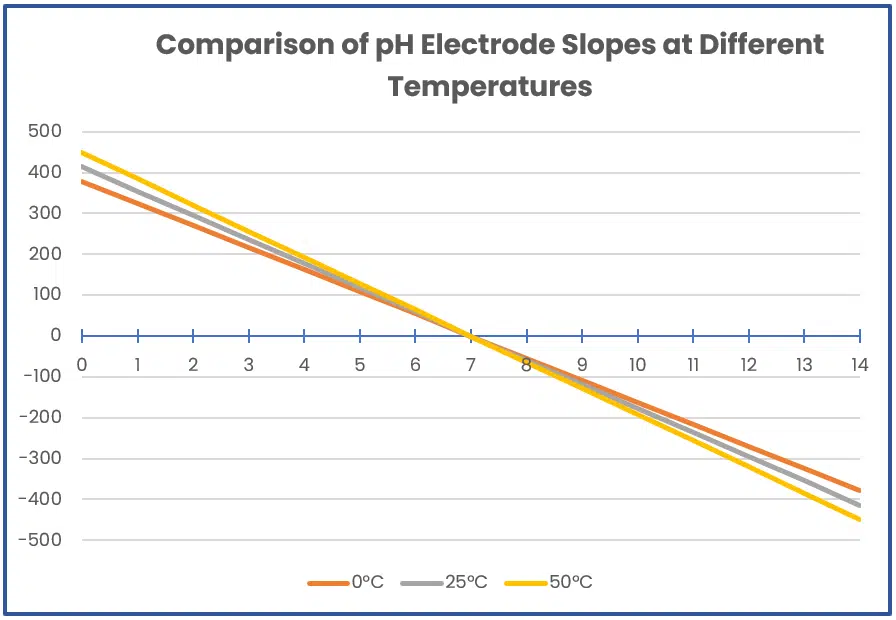
Image 3. Graph showing how pH electrode slope changes with temperature.
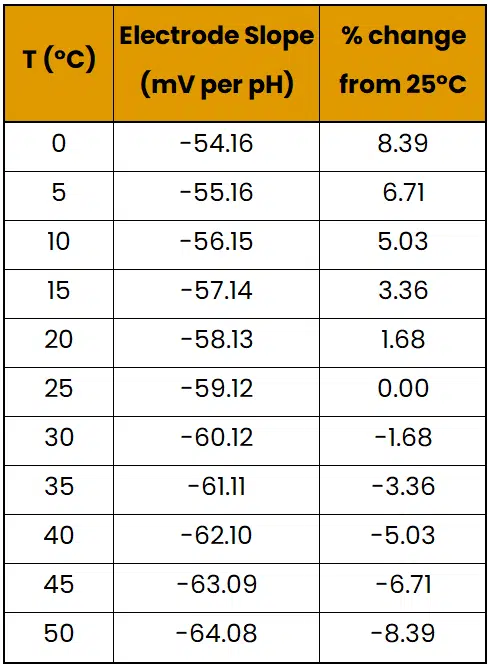
Image 2. Table showing how pH electrode slope varies with temperature.
As shown, temperature changes could result in a needless loss of accuracy of over 8% if not accounted for. While it is possible to collect temperature with each pH measurement and adjust the slope manually, many pH sensors include a temperature sensor to automatically adjust their output. This feature is called Automatic Temperature Compensation (ATC) and saves the user a significant amount of work.
While ATC significantly increases the accuracy of the sensor, always remember to also do the following:
- Use an electrode kept in good condition.
- Calibrate the sensor in buffers close to the same temperature as the sample.
These simple steps ensure accurate measurements for most applications.
To be clear, ATC ensures accurate calibration and measurement of pH over a range of temperatures because it corrects for physical changes in the sensor. It does not correct for any pH shifts in the sample itself. As such, solution temperature compensation is required when comparing two pH measurements at different temperatures. Why this is and how to compare pH samples at different temperatures is explained in the next section.
How Temperature Affects pH
Pure water under standard conditions (25°C and 1 atm) is 7 pH. At 30°C, the pH drops to 6.92. This is not an error. Water dissociates into ions more at higher temperatures, leading to a higher concentration of hydrogen ions (or H3O+). A higher concentration of H3O+ ([H3O+]) results in lower pH, but it does not mean the sample has changed acidity. Here’s why: Based on the Bronsted-Lowry acid-base theory, acids and bases are defined by their interactions together, expressed in the following chemical equilibrium equation: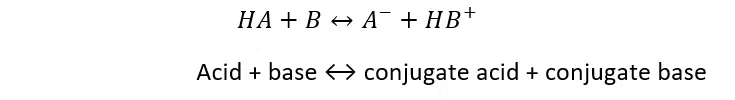
Image 4. Bronsted-Lowry acid-base equilibrium equation.
The ratio of products to reactants is expressed as an equilibrium (or dissociation) constant (K). This constant is affected by temperature, pressure, and ion concentration of water, with temperature having the largest effect.
Water can give and receive protons, allowing it to act as both an acid or base for other compounds, or even for itself in a process called autoprotolysis. The equilibrium equation is provided below:
Image 5. Water autoprotolysis.
Our definition of acids and bases normally relies on referencing the pH scale, with 7 pH in standard conditions being neutral, meaning the solution has an equal ratio of H3O+ : OH–. Below 7 pH is acidic, meaning there is more H3O+ than OH–, and vice versa above 7 pH. Outside of standard conditions, these assumptions no longer apply. Pure water at 30°C has more H3O+ than at 25°C so pH is lower, but an equal amount of OH– is also present, so the neutral pH point shifts to 6.92. As such, acidity does not change.
The graph below provides the equilibrium constant of pure water (Kw) at different temperatures and demonstrates the corresponding shift of neutral pH.
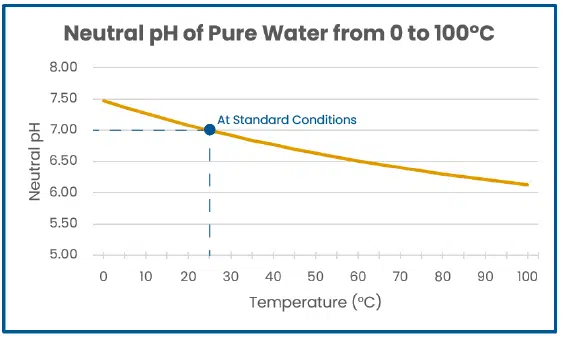
Image 6. Graph of neutral pH change with temperature.
Again, this pH change is not a measurement error, but it prevents accurate acidity comparison of samples at different temperatures. To do so, the equilibrium constant of the solution must be known.
Samples may have different chemical or biological reactions to changing temperatures so the equilibrium constant of each solution must be determined empirically by heating or cooling a representative sample across the temperature range of interest. Only after the equilibrium constant of a sample is known can two pH measurements from different temperatures be compared at a reference temperature.
Solution temperature compensation is not required or realistic for most environmental and wastewater applications because temperature normally does not change more than ±10°C so only a ~2% pH difference may be introduced for natural water. This assumption may not hold true for chemical- or heat-intensive wastewater processes, however, so it is important to be aware of.
Conclusion
The effects of temperature on pH can be significant so it is important to compensate for them. While many sensors come with automatic temperature compensation, errors can still occur due to improper calibration so always have the sample solution and sensor at the same temperature. In addition, avoid comparing samples at different temperatures unless the measurements are solution temperature compensated. Following these tips significantly increases the accuracy and ensures data is analyzed properly.
If you need a pH sensor that works anywhere and anytime with little maintenance, then the pHionics STs Series pH sensor is perfect for you. A large, gel-filled, double junction reference electrode ensures stable measurements over long periods of time. In addition, the sensor is narrow, fully submersible, and made of chemical-resistant materials for many years of reliable service. Click here to visit our pH sensor product page and learn about more if its features.
Thank you for reading. Please reach out to us if you have any questions or feedback on the article.
Suggest an Article or Video Topic!
Recent Articles

How Glass pH Electrodes Work
In this video, learn about the electrochemistry that allows silver/silver chloride pH electrodes to measure the acidity of solutions. Modern electrode design is also reviewed to demonstrate what improvements have been made and what weaknesses are still present. Click...
STs Series Sensor Storage
STs Series sensors are constructed from high-quality, durable materials that can be stored for long periods of time. The only weak point is the electrode, which can be damaged or expire during storage in the wrong conditions. These conditions vary depending on the type of electrode, which is why we have different storage instructions for each sensor.
How a Glass pH Electrode Works
A comprehensive article covering how glass electrodes measure pH in a simple, understandable format. Specifically for silver/silver chloride electrodes.
