The Best Storage Solutions for pH Sensors
This article is specifically for pH sensors with glass electrodes and silver/silver chloride references.
What is the purpose of storage?
To put it simply, the purpose of storage is to protect the accuracy and stability of the sensor until it needs to be used. Improper storage reduces the lifespan of your electrode and prevents you from getting the full value out of it.
What are the limiting factors for accuracy and stability of pH sensors?
- The reference electrolyte, which is neutral pH, 3.0M KCl for the pHionics instruments and most of the industry. As soon as the electrolyte is either depleted or contaminated, the sensor loses a stable reference and cannot hold calibration.
- The sensing element, which for pH electrodes is a hydrogen-ion-sensitive hydrated gel layer on the electrode bulb that must be kept wet.
Let’s look at a few different examples of how storage conditions affect the limiting factors and, therefore, the lifespan of pH sensors.
Option 1: Storing a pH sensor without storage solution
As the reference electrolyte is exposed to the environment through an outer junction, it evaporates over time and the bulb dries out. The electrolyte dries out within a few months and the electrode stops working, resulting in wasted money spent on a replacement electrode.
If the sensor is pulled from storage before the electrolyte dries out or if the electrolyte is refilled, then you may attempt to use it and find the measurements drift or stabilize very slowly. This is a result of the bulb drying out. Our engineers have successfully recovered some electrodes by soaking them in 3.0M KCl overnight after drying out, but results are inconsistent.
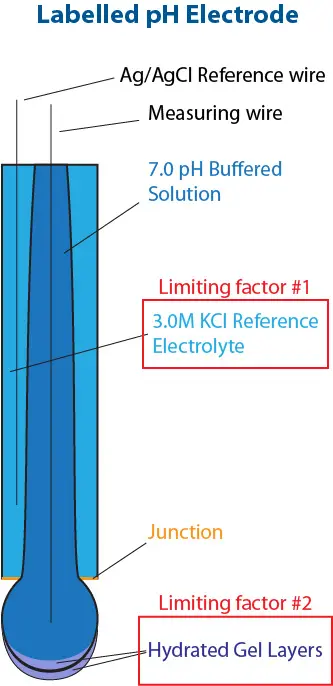
Image 1. Labelled Glass pH Electrode
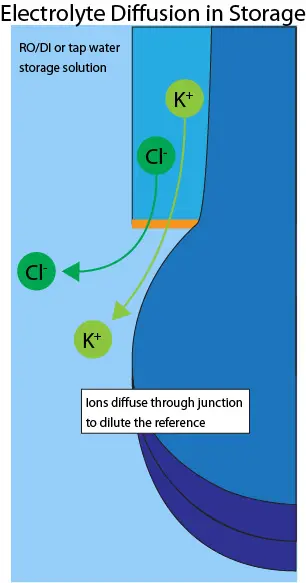
Image 2. Example of ion diffusion caused by storage of electrode in tap water
Option 2: Storing a pH sensor in RO/DI or tap water
With this option, the reference electrolyte and H+-sensitive gel do not dry out. The potassium and chlorine ions diffuse slowly through the junction and into the water until an equilibrium is reached, resulting in a diluted reference (shown in Image 2.). Storing a pH sensor in tap water or, worse, RO/DI water, is the same as having it in use, so the time in storage results in an equivalent decline in electrode lifespan. If left for long enough, the reference becomes unstable and no longer holds calibration, requiring the electrode to be replaced.
Option 3: Storing a pH sensor in reference solution (3.0M KCl for most products. Check with your manufacturer)
This option has the same benefit as Option 2, as the reference electrolyte and gel do not dry out. In addition, the internal reference solution and storage solution are the same so there is no diffusion potential pulling out the potassium and chlorine. The electrode is well preserved and does not have its usable lifespan reduced.
One concern worth mentioning is that a build-up of salt crystals may occur after long periods of storage, resulting in slow response times. These crystals can be removed with a soft cloth/brush or by soaking in 0.1M HCl for up to 20 minutes.
Conclusion
Always store a pH sensor with the electrode submersed in the same solution as the reference electrolyte solution. If it is not available, a solution of similar ionic content and neutral pH should be used in the meantime.
If you are looking for a pH sensor that can last for a long time, then the pHionics STs Series pH is perfect. We have combined a fast, accurate, reliable electrode (average lifespan of 3 years) with body made of high quality materials that typically last 10+ years. The STs Series pH also contains an isolated, 2-wire, 4-20 mA transmitter for excellent noise-resistance and safety. Find out more by visiting the STs Series pH product page.

Check out our video for a visual demonstration of how improper storage affects pH electrode life.
Suggest an Article or Video Topic!
Recent Articles

How Glass pH Electrodes Work
In this video, learn about the electrochemistry that allows silver/silver chloride pH electrodes to measure the acidity of solutions. Modern electrode design is also reviewed to demonstrate what improvements have been made and what weaknesses are still present. Click...
STs Series Sensor Storage
STs Series sensors are constructed from high-quality, durable materials that can be stored for long periods of time. The only weak point is the electrode, which can be damaged or expire during storage in the wrong conditions. These conditions vary depending on the type of electrode, which is why we have different storage instructions for each sensor.
How a Glass pH Electrode Works
A comprehensive article covering how glass electrodes measure pH in a simple, understandable format. Specifically for silver/silver chloride electrodes.
