pH Electrode Guide
Table of Contents
There are two different methods of measuring pH currently available called potentiometric and photometric, with several different subsets of technologies for each. This article focuses on the real-time monitoring capabilities of glass pH electrodes used in industrial process control, wastewater treatment, and environmental data logging and provides comparisons of common features to assist in purchasing the right electrode/sensor for your application.
Methods of pH Measurement
First is photometric measurement, which uses pH–sensitive indicators added to solution and evaluated either by eye or spectrophotometer. Photometric is cheap but has slow response times, making it a great option for occasional measurements but samples must be obtained and prepared and results are heavily affected by the color of the sampled solution. An operator must always be present to obtain results which limits the range of applications. Real-time information capture is not possible so the photometric method is not useful to notify plant operators or scientists of sudden changes that may be disastrous in production processes or experiments.
Potentiometric measurements are broken down into two types: glass and ion-sensitive field-effect transistors, or ISFETs. These forms of measurement use pH-sensitive glass or silicon chips, respectively, to create and measure the difference in potential energies between a reference and test solution. They have high accuracy and can transmit data autonomously in real-time, making them perfect for applications such as process control or environmental datalogging.
Comparison of Potentiometric Methods
Glass Electrode Anatomy
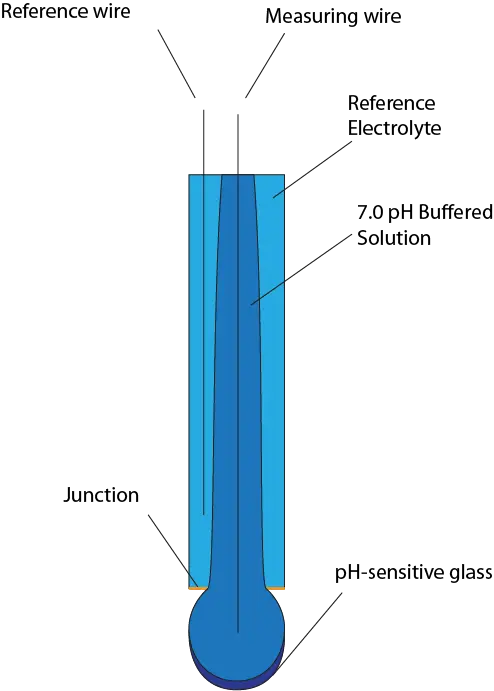
Glass Electrodes
A glass pH electrode uses a bulb made from pH-sensitive glass to measure a voltage when the electrode is submersed (Details of how it works will be found in the next section). This method is highly accurate, has an excellent response-time, and can be adapted to fit most applications. The downsides are:
- The glass is fragile if not properly protected
- The reference solution becomes contaminated over time and accuracy decreases
- The reference wire acts as an antenna for any electrical charge in the solution. (This is solved by differential amplification and isolation)
ISFET Electrodes
Ion-sensitive field-effect transistors, or ISFETs, are electronic devices using silicon chips to detect voltage potential between the sample and the semiconductor material. They are exceptionally durable as there are no exposed glass pieces. That feature, combined with easy sterilization processes, is why they are commonly used in food processing. Unfortunately, they are more expensive than potentiometric devices, less accurate, and have lower long-term stability when compared to potentiometric techniques, as well as a few other issues which make them unsuitable for many industrial and environmental applications. A good comparison can be found here.
How Glass Electrodes Work
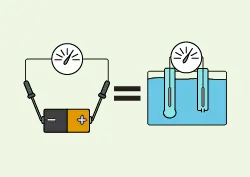
Just as a voltmeter measures the difference in energy potential between two points in a circuit, pH electrodes measure the difference in energy potential between two solutions. Visually, the two systems may look quite different but, in their basic forms, they are both just two wire leads contacting different points or solutions.
When measuring the voltage of a battery, a lead is placed on the positive and negative ends to form a circuit. This allows a small number of electrons to flow from the point of higher potential energy to the point of lower potential energy, which creates a magnetic field that moves the needle of an analog voltmeter. The magnetic field strength is proportional to the voltage.
Voltmeters can simply measure the voltage that is present in a charged battery. In contrast, two solutions do not normally have potential energy built up between them so a pH electrode must find a way to “charge” the solutions.
To create a potential gradient and “charge” the solution, glass sensitive to hydrogen ions is used to form the bulb. Thin hydrated layers form where the glass contacts solution. If there is a difference between the number of hydrogen ions on one side of the glass, then a gradient with measurable voltage is formed.
As the diagram to the right shows, the direction of current flow changes based on which side of the pH-sensitive glass has the highest concentration of H+ ions. Each 1 pH shift below or above 7 pH generally represents a +59.16 mV or –59.16 mV respective change based on the Nernst Equation, with 0 pH yielding approximately +414 mV, and 14 pH providing about -414 mV.
The two wires are in separate chambers known as half-cells (One half-cell is light blue and the other is dark blue). They are made of materials with redox potential to allow for charge to flow, with the most common being silver wire plated with silver chloride and surrounded by concentrated KCl. When potential builds up between the glass bulb, a small amount of charge flows because the wires can either accept or give up electrons, as shown in the equation below:
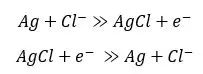
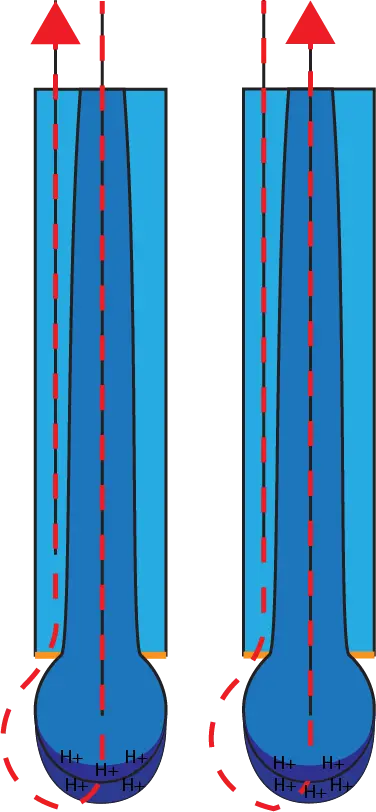
Click here for a more complete explanation of how pH electrodes work.
How Electrode Junctions Work
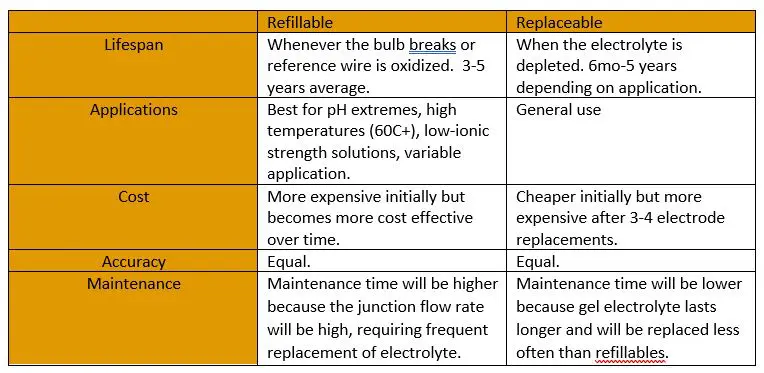
What connects the two wires together is a liquid junction (Shown in orange on the diagrams). This junction is made of a porous material that allows a small volume of reference solution to flow out of the reference half-cell, essentially acting as a liquid wire. It achieves the difficult goal of isolating the reference solution from the sample solution while still allowing current flow.
Junctions are designed to allow outflow of reference electrolyte to form a circuit and keep the junction unclogged, but they also allow inflow of the solution being measured. Some of the sample solution will enter the reference cell and dilute the potassium chloride or react with the silver elements, respectively resulting in drift over time or a dead electrode. While the electrode must be replaced if the silver reference wire is attacked by cyanides or sulfites, etc., dilution and depletion of the reference electrolyte depends on the ionic strength and pH of solution and is typically solved in one of two ways: refillable or replaceable electrodes.
Comparison of Refillable vs Replaceable Electrodes
One method is to use refillable electrodes, which have a hole that can be opened or closed to add or change electrolyte as needed. The second method is using replaceable or sealed electrodes, which are often filled with electrolyte gels or polymers instead of solution, giving them a lower flow rate. Typical replaceable electrode life is about 2-3 years but depends heavily on how quickly the reference solution is diluted. They will not need to be replaced as often as refillable electrodes must be refilled so they are considered lower maintenance and are generally cheaper than their refillable counterparts. Accuracy can be as high as ±0.01-0.02 pH for both types so the main reason to use refillable electrodes is for samples at extremes (e.g. above gel/polymer melting point, or in strong acids, bases, or low-ionic strength solutions where dilution occurs rapidly). Be aware that refillable electrodes will also last less time in these extreme situations than it otherwise would because the reference wire will become oxidized over a shorter period.
Solving Reference Electrolyte Dilution
An effective method to extend the life of the reference lead wire is to add a second junction to the reference half-cell, with these types of electrodes being called…
Wait for it…
DOUBLE JUNCTION.
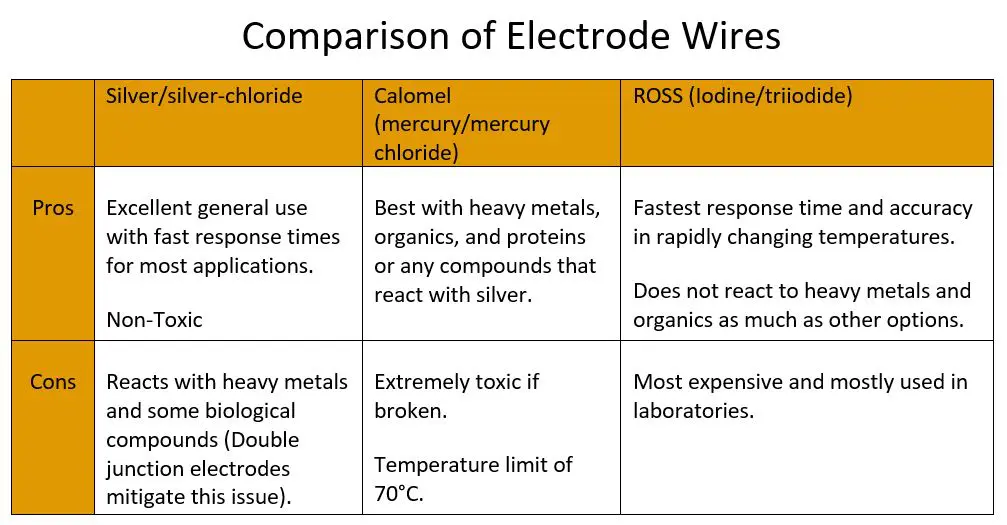
While the name might not be clever, the design is extremely effective. A second, inner chamber is created that houses the reference wire, resulting in significantly longer diffusion times across a second membrane and reducing the oxidation rate of the wire element. This has resulted in greatly increased life expectancy and a wider range of applications for electrodes with double junctions. There is no case where single junction outperforms double junction, but single junction electrodes are slightly cheaper. Double junctions have also made Calomel electrodes effectively obsolete as nearly all the silver chloride of Ag/AgCl electrodes remains in the inner chamber. Calomel will still perform better in solutions with high concentrations of heavy metals, proteins, and some organic matter, but the risk of mercury poisoning and contamination due to breakage often outweighs the benefits in performance.
Electrode Bulb Shape
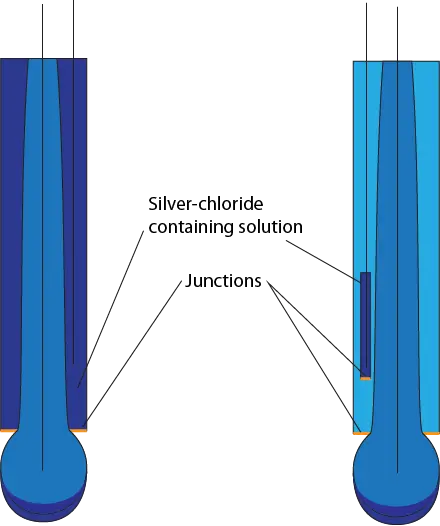
The final consideration for electrode compatibility with your desired application is the shape of the pH-sensitive glass. The most common shape is a bulb, but different shapes exist to improve contact of the electrode with the solution being measured. See the following chart for a breakdown of uses for each shape.
Conclusion
Let’s review important aspects of a pH electrode by considering the electrode pHionics uses in our STs Series pH sensor and transmitter. Our electrode is silver/silver chloride, which avoids the toxicity of Calomel (mercury/mercury chloride) reference leads but makes our electrode vulnerable to reaction with heavy metals, sulfur, and proteins. A Teflon double junction significantly reduces the rate at which these compounds enter the reference solution and contact silver, thereby increasing electrode life in applications containing high concentrations of heavy metals etc. Our electrodes are gel-filled and replaceable, meaning that they have a low electrolyte flow rate and are low maintenance but will only withstand up to 70°C. Lastly, they incorporate a bulb style electrode tip, making it applicable to any application that does not require flat-surface measurement or piercing.
The information in this article represents only the tip of the iceberg with respect to the field of pH electrode studies, but hopefully we helped clarify some of their important features. While electrodes plays a large part in application compatibility, the sensor and transmitter are also important so always remember to do your research.
pHionics is here to provide information and advice on water quality systems so please don’t hesitate to reach out with any questions.
Suggest an Article or Video Topic!
Recent Articles

How Glass pH Electrodes Work
In this video, learn about the electrochemistry that allows silver/silver chloride pH electrodes to measure the acidity of solutions. Modern electrode design is also reviewed to demonstrate what improvements have been made and what weaknesses are still present. Click...
STs Series Sensor Storage
STs Series sensors are constructed from high-quality, durable materials that can be stored for long periods of time. The only weak point is the electrode, which can be damaged or expire during storage in the wrong conditions. These conditions vary depending on the type of electrode, which is why we have different storage instructions for each sensor.
How a Glass pH Electrode Works
A comprehensive article covering how glass electrodes measure pH in a simple, understandable format. Specifically for silver/silver chloride electrodes.
